
The remaining c-axis is more variable (13–23 Å). There is always a second axis of 9.5–9.6 Å in length, which, in order to facilitate comparison between the different structures, is here always taken to be the a-axis. All structures are monoclinic and have a b-axis close to 5.2 Å. Data integration and cell refinement were performed with SAINT-Plus (Bruker, 2014 ) with subsequent absorption correction by SADABS (Bruker, 2014 ) and structure solution with SHELXLT (Sheldrick, 2015 a ). Data sets were collected on cooling and heating from room temperature, except that data for L-Nle at 180 K were collected after first cooling the crystal to 100 K.
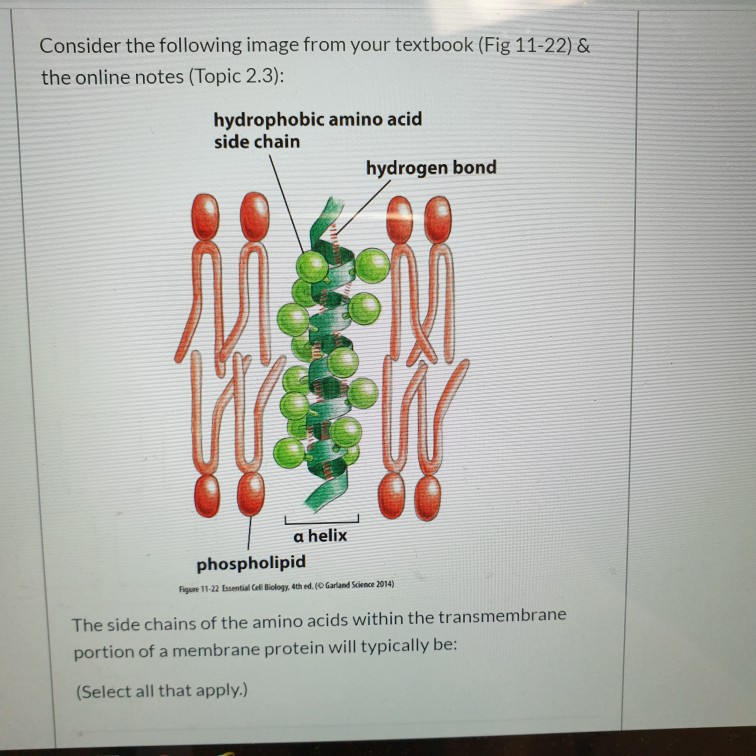
Do hydrophobic amino acids hydrogen bond software#
Single-crystal X-ray data collections were carried out with APEX2 software (Bruker, 2014 ) on a D8 Venture single-crystal CCD diffractometer equipped with an Oxford Cryosystems Cryostream Plus cooling unit and Mo K α radiation ( λ = 0.71069 Å). X-ray data collection and structure solution L-Nva has not been subject to X-ray diffraction investigations in the past.Ģ.4. Only a single structure (at room temperature) is available for L-Nle (Torii & Iitaka, 1973 ). For L-Abu we previously found two forms at 110 K, both with four molecules in the asymmetric unit ( Z′ = 4 Görbitz, 2010 ).
Only one crystal form is known for L-Met, which has been studied at room temperature (Torii & Iitaka, 1973 ), 150 K (Sadler et al., 2005 ) and 120 K (Dalhus & Görbitz, 1996 a ). Little is known by comparison about the corresponding enantiomeric amino acids.
Do hydrophobic amino acids hydrogen bond series#
A series of unique solid-state phase transitions, involving sliding along the hydrophobic interfaces between adjacent bilayers, have been observed for DL-methionine (Met Mathieson, 1952 Taniguchi et al., 1980 Alagar et al., 2005 Görbitz et al., 2014, 2015 Görbitz, 2014 ), DL-2-aminobutyric acid (Abu Görbitz et al., 2012 Nakata et al., 1980 Voogd & Derissen, 1980 Akimoto & Iitaka, 1972 Ichikawa & Iitaka, 1968 ), DL-norvaline (Nva Görbitz, 2011 ) and DL-norleucine (Nle Coles et al., 2009 Harding et al., 1995 Dalhus & Görbitz, 1996 b Smets et al., 2015 van den Ende & Cuppen, 2014 ) as well as for the four quasiracemates L-Nva: D-Nle, L-Nva: D-Met, L-Nle: D-Met (Görbitz & Karen, 2015 ) and L-Abu: D-Met (Görbitz et al., 2016 ). The crystal structures of amino acids with hydrophobic side chains invariably incorporate molecular bilayers, each with a hydrogen-bonded core and two surfaces shaped by the amino acid side chains. An accurate refinement now reveals extensive disorder for both molecules in the asymmetric unit, while two previously unknown phases occur above room temperature.

L-Methionine has previously been described as being fully ordered at room temperature. Ordering of L-norleucine upon cooling even proceeds via an incommensurately modulated structure. The multiple transitions between them involve a number of different processes: alteration of the hydrogen-bond pattern, to our knowledge the first example of this observed for an amino acid, sliding of molecular bilayers, seen previously only for racemates and quasiracemates, concerted side-chain rearrangements and abrupt as well as gradual modifications of the side-chain disorder. Unlike other enantiomeric amino acids investigated until now, this group featuring linear side chains displays up to five distinct phases. The solid-state phase transitions and intermediate structures of S-2-aminobutanoic acid ( L-2-aminobutyric acid), S-2-aminopentanoic acid ( L-norvaline), S-2-aminohexanoic acid ( L-norleucine) and L-methionine between 100 and 470 K, identified by differential scanning calorimetry, have been characterized in a comprehensive single-crystal X-ray diffraction investigation.
